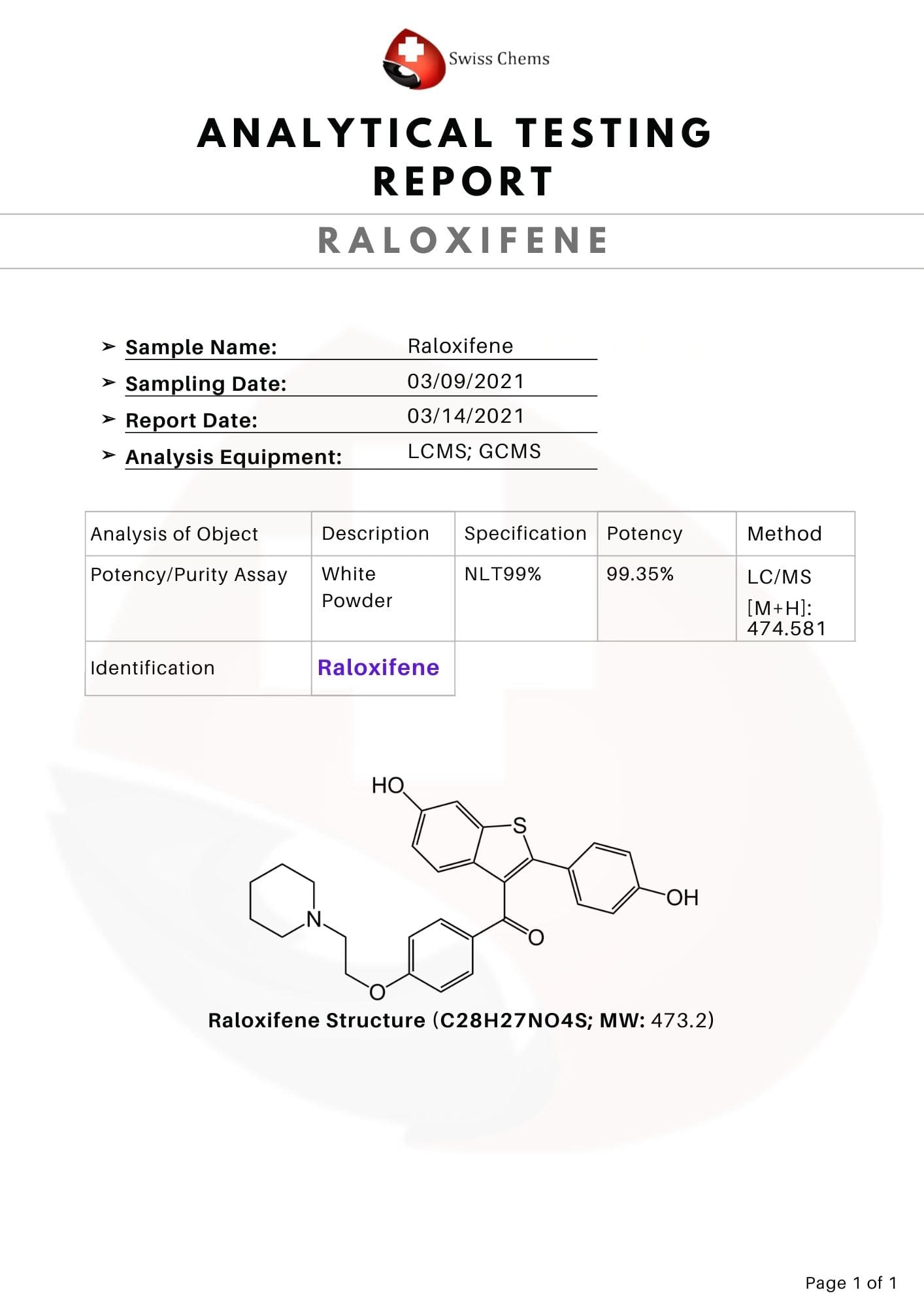
Raloxifene, developed in 1997 and sold under the brand name Evista, among others, is a medication used to reduce breast cancer risk in postmenopausal women with osteoporosis. Raloxifene was first presented in 1997 to help with post-menopausal osteoporosis and was subsequently approved in 1999. Its effect on the reduction on breast cancer risk was also accepted in 2007.
Raloxifene capsules is indicated for the treatment and anticipation of osteoporosis in postmenopausal ladies. It is also used for the treatment and reduction of risk of invasive breast cancer. Raloxifene has been found effective to combat gynecomastia.
Raloxifene tablets is contraindicated in lactating and pregnant women or women suspecting pregnancy.
It also may be a concern to women with active or past history of venous thromboembolic events including deep vein thrombosis, pulmonary embolism, and retinal vein thrombosis. You should never take more dosage than the doctor prescribes.
Other Names: Keoxifene; Pharoxifene; LY-139481; LY-156758; CCRIS-7129
Total Amount of the Active Ingredient: 1200 mg
Quantity: 60caps
Packaging: capsulated, bottled
Label:L: 14cm X W: 5,7cm
Shipping:
•USA
•Canada
•International
•Europe
•South Asia: (Afghanistan, Bangladesh, Bhutan, India, Maldives, Nepal, Pakistan, Sri Lanka)
•Middle Eastern: (Egypt, Bahrain, Cyprus, Iran, Iraq, Israel, Jordan, Kuwait, Lebanon, Oman, Qatar, Saudi Arabia, Syria, UAE, Yemen, Turkey)
If your shipment was seized (International Orders), we will provide a 50% discount applicable on your next purchase. Please contact us for more information.
Product made and housed in the USA and Europe.
Product Quality Guarantee
All of our products are lab tested and the results are occasionally published on the website.
You can have the product you bought from us tested at any HPLC licensed testing facility and if the results are negative, we will refund the following:
- Cost of HPLC test
- Total amount of the order shipping fee
Additional information
| Weight | 3 kg |
|---|


Reviews
There are no reviews yet.